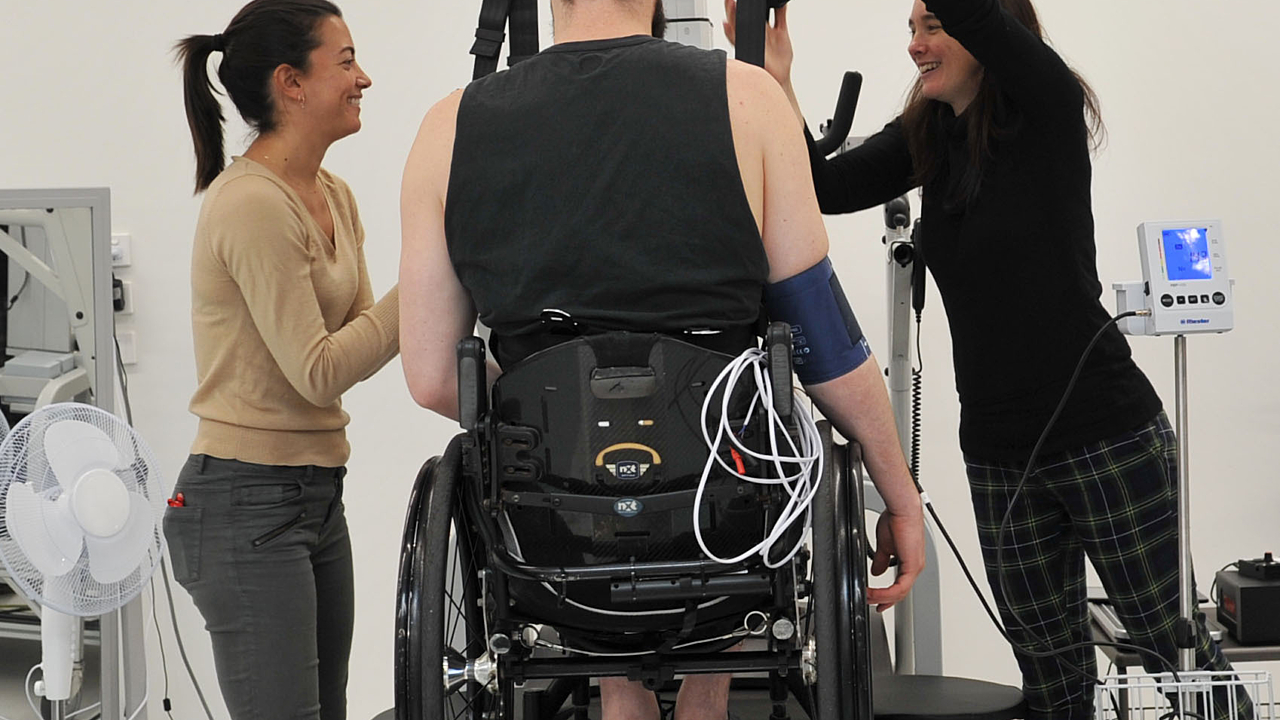
Australian researchers are embarking on a landmark international trial which aims to help people with spinal cord injury walk again.
Led by researchers at Neuroscience Research Australia (NeuRA) and primarily funded by SpinalCure Australia and CatWalk Trust NZ, the eWALK trial aims to harness the power of neuroplasticity to restore the function of remaining spinal nerves.
The therapy, neurostimulation, sends electrical impulses through electrodes that sit on the surface of the skin over the spinal cord. When it is coupled with step and walking training in people with chronic paraplegia, the therapy helps to rewire the neural pathways that have been impaired.
The eWALK trial emerged from a community initiative spearheaded by SpinalCure Australia. Thanks to their support, NeuRA has now started testing this innovative technology in a randomised controlled trial lead out of Sydney, with sites in Chicago, Glasgow and Toledo.
“Our scientific panel chose to fund this research because neurostimulation holds the most promise out of current research to bring tangible improvement to people with spinal cord injury in the near future,” said SpinalCure Australia CEO, Kathryn Borkovic.
“Professor Simon Gandevia at NeuRA is one of the world’s leading researchers in the field of human movement disorders, and NeuRA has both an excellent reputation for quality research and is a world-renowned independent not-for-profit research institute,” said Ms Borkovic.
Study lead for eWALK and Deputy Director of NeuRA Professor Simon Gandevia says the results of previous small neurostimulation studies are compelling and positive, but a lack of highly controlled clinical trials has prevented this technology from being converted into clinical practice.
“Neurostimulation is like a hearing aid for the spinal cord. The idea is that tailored electrical currents can amplify messages transmitted via surviving neural pathways, thereby enhancing communication between the brain and the body.
“Neurostimulation restores meaningful movements that would be otherwise impossible. Volunteers in research overseas have even been able to stand and walk. However, until now, researchers have not been able to test this promising technology in a clinical setting,” said Professor Gandevia.
With more than 20,000 Australians living with paralysis, bearing a cost of $3.7 billion each year to the economy, SpinalCure Australia and NeuRA are calling on the Government and community to provide financial support to extend this crucial research.
Ms Borkovic says at least another $20 million is needed to expand the eWALK trial to focus on a wider range of spinal injuries and other bodily functions that matter to people living with spinal cord injury.
“The impact of a spinal cord injury is life-shattering, and there is no treatment, pill or lifestyle change that can mitigate its effects. Loss of movement is just the tip of the iceberg – ongoing pain, digestive health issues, pressure sores, spasm, loss of bladder and bowel control and impaired sexual function are just some of the effects that can make everyday life so difficult.
“Research into spinal cord injury treatments is essential to improve the outcomes for people living with spinal cord injury.
“We need to fund research like this because science is showing that huge improvements in function are possible and can increase people’s independence and wellbeing.
“SpinalCure Australia, together with CatWalk Trust, are so proud to have funded the eWALK trial to date. But we need more funds to expand and assess the potential of this therapy to help people living with spinal cord injury more broadly,” said Ms Borkovic.
The eWALK trial is funded by SpinalCure Australia and CatWalk Trust, with additional funding from Spinal Cord Injuries Australia. Initial results are expected in around 18 months.
To make a donation, please visit spinalcure.com.au or to find out more about the eWALK trial, please visit neura.edu.au.